Calculate the pH of a solution that is 0.240 mol L-1 in nitrous acid ( 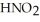 ) and 0.320 mol L-1 in potassium nitrite (
) and 0.320 mol L-1 in potassium nitrite ( 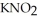 ) . The acid dissociation constant of nitrous acid is 4.50 ×
) . The acid dissociation constant of nitrous acid is 4.50 ×  .
.
Definitions:
Resting State
A period of mental and physical inactivity during which an individual's consciousness is engaged but not focused on the external environment, often used in neuroimaging studies.
Action Potential
A rapid rise and subsequent fall in voltage or membrane potential across a cellular membrane, vital for nerve impulse transmission.
Synaptic Vesicles
Small sacs within a neuron that store neurotransmitters, which are chemicals that transmit signals across synapses.
Synaptic Knob
The bulbous end of a presynaptic neuron's axon terminal that houses neurotransmitters and is responsible for transmitting signals to other neurons at the synapse.
Q10: Calculate the K<sub>sp</sub> for silver carbonate if
Q30: Use the provided reduction potentials to calculate
Q43: Identify the expression for the solubility product
Q53: Which of the following metals will dissolve
Q56: Determine the pH of a 0.62 mol
Q63: A 100.0 mL sample of 0.10 mol
Q65: Given that E°<sub>red</sub> = -1.66 V for
Q84: Identify a homogeneous catalyst.<br>A) SO<sub>2</sub> over vanadium
Q84: Calculate the cell potential for the following
Q109: Which of the following is the correct