Determine the enthalpy change for the decomposition of calcium carbonate
CaCO3(s) CaO(s) + CO2(g)
Given the thermochemical equations below.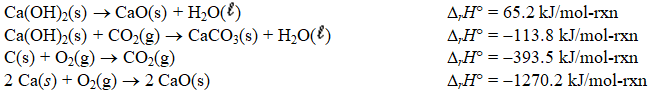
Definitions:
Women's Experience
Women's experience pertains to the particular conditions, events, and perspectives that shape the life and identity of women, taking into account the diversity and complexity of their roles in society.
Profitable Deviance
Involves acts or practices that, while deviating from societal norms or laws, result in financial gain for individuals or corporations.
Deviant Opportunity
Circumstances that encourage or allow individuals to engage in actions that deviate from societal norms or laws.
Deviant Motivation
The drive or incentive behind actions that deviate from societal norms or expectations.
Q18: What is the net ionic equation
Q27: A point in a standing wave that
Q32: The standard molar enthalpy of formation of
Q38: As pure molecular solids,which of the following
Q42: The ground-state electron configuration of a <img
Q55: How much energy is gained by
Q60: Order the four metric units provided from
Q64: Which of the following would
Q77: An atom of which of the following
Q91: A balloon is filled with He