Consider the Following Reaction 3Fe(s) + 2Al3+(aq)
This Reaction Takes Place in the Electrochemical
Consider the following reaction.
3Fe2+(aq) + 2Al(s) 3Fe(s) + 2Al3+(aq)
This reaction takes place in the electrochemical cell shown below. 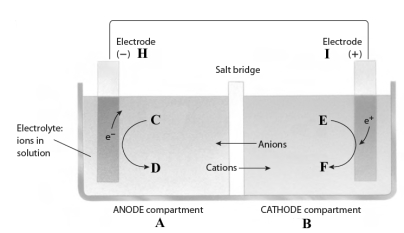
Complete the following questions using the letters shown in the image.
-The electrons flow from electrode _____ to electrode _____.
Definitions:
Unlimited Capacity
A theoretical condition where a system can handle any amount of workload without restrictions or limitations.
Gantt Load Chart
A Gantt Load Chart is a visual project management tool used to schedule and track the progress of tasks over time, often displaying the start and end dates of tasks in a project and how they overlap.
Loading Time
The time period required to prepare and load goods into transport vehicles or machinery, critical in logistics and manufacturing for determining overall efficiency and productivity.
Idle Time
The time during which resources, such as machinery or labor, are not being used for production, often due to lack of orders or maintenance.
Q6: Of the following, which is the strongest
Q6: The use of a fire blanket in
Q12: Commercial nitrogen is produced from the air
Q16: Digestion converts carbohydrates into<br>A) amino acids.<br>B) fatty
Q20: In the balanced equation for the combustion
Q21: Which substance is grain alcohol?<br>A) methanol<br>B) ethanol<br>C)
Q25: Chemical equilibrium exists for a chemical reaction
Q55: The following structure would be classified as
Q78: How many resonance structures can be drawn
Q81: Rutherford's foil experiment provided evidence for what